 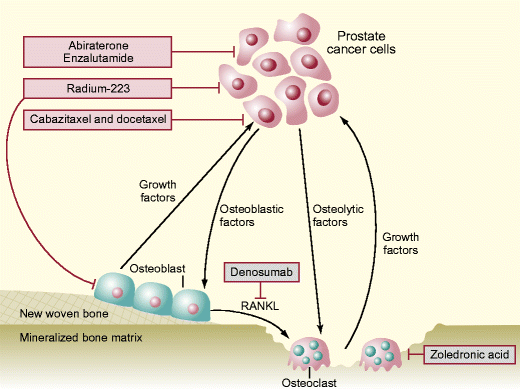
When used as first-line treatment of patients with mCRPC and bone or visceral metastases, enzalutamide improved OS by 2 months compared with placebo (32.4 vs 30.2 months P<0.001). 7, 8 The second one is enzalutamide, an androgen receptor antagonist. Its approval relied on a 4-month OS improvement in patients with bone metastatic prostate cancer after docetaxel treatment compared with placebo (15.8 months vs 11.2 months P<0.0001) and also in chemotherapy-naive patients (34.7 vs 30.3 months P=0.0033). The first one is abiraterone acetate (AA) that targets CYP17A1, a key enzyme involved in androgen synthesis. 6 The last two drugs are “second generation” hormonal treatments that target the androgen receptor signaling pathway. 5 The same year, it was shown that sipuleucel-T, an autologous cellular immunotherapy, prolongs survival in chemotherapy-naive patients with asymptomatic or minimally symptomatic mCRPC compared with controls (25.8 months in the sipuleucel-T group vs 21.7 months in the placebo group). 3, 4 Then, in 2010, the results of the TROPIC study in a post-docetaxel setting (OS increase of 2.4 months compared with mitoxantrone 12.7 vs 15.1 months P=0.0001) led to the approval of cabazitaxel, a taxane with lower affinity for drug efflux pumps compared with previous molecules of the same class. First, docetaxel, a microtubule poison from the taxane family, was approved on the basis of a 2.5-month survival improvement (16.4 vs 18.9 months P=0.009) compared with mitoxantrone (standard treatment). Since 2004, five drugs have been approved for mCRPC treatment, leading to an improvement of progression-free survival and OS. Finally, it will give information on the ongoing trials that assess 223Ra for prostate cancer management.īesides 223Ra, several other agents have shown efficacy in metastatic CRPC (mCRPC). It will then summarize the available clinical data and the place of 223Ra in the current clinical practice. It will first describe the mechanism of action of this new agent against bone metastases. This review, which is the result of a multidisciplinary collaboration by the Intergroupe Coopérateur Francophone de recherche en onco-urologie (ICFuro), discusses the place of 223Ra in the therapeutic landscape of prostate cancer. 2 This led to its approval by the US Food and Drug Administration in 2013. In the Phase III trial ALSYMPCA, 223Ra showed an overall survival (OS) benefit in patients with castration-resistant prostate cancer (CRPC) and symptomatic bone metastases. Therefore, it specifically targets bone metastases. Radium-223 ( 223Ra Xofigo ®) is an α-emitting radionuclide that, like calcium, is incorporated in the bone matrix at sites of active mineralization via osteoblasts. Bone metastases often lead to pain or skeletal events (fracture, spinal cord compression) and, therefore, may decrease the patients’ quality of life. 1 Although most patients are cured by local treatment, 20%–30% will have a recurrence, especially in bone. Prostate cancer represents the second most frequent cancer worldwide, with an incidence of 1.09 million patients in 2012.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
